Frost Closes SciVac Deal, A Potential Blockbuster Lineup in Hepatitis?
Towards the end of last year, we got word that Dr Phillip Frost, chairman and CEO of Opko Health, Inc. (NYSE:OPK), was looking to restructure then private company SciVac (now SciVac Therapeutics Inc. (SVACF) into a public entity. Little more was heard of the matter until March 2015, when gold and precious metals exploration company Levon Resources Ltd announced that it had entered into an agreement pursuant to which Levon would acquire 100% of the issued and outstanding ordinary shares of SciVac. OPKO had a 45% stake in SciVac, and so the deal would see current SciVac owners take 68.4% of the outstanding shares of the new company after its close, and Opko take a little over 30%. At first glance the arrangement can seem a little bit convoluted, but a closer look reveals it is very similar in structure to a number of other public shell spins Frost has orchestrated in the past.
On July 10, SciVac announced the completion of the transaction, and its granting of approval by the Toronto Stock Exchange. Frost has a dedicated following of both individual and institutional investors that monitor his activity, so any stock in which he has a hand has the potential to revalue quickly on positive news. Further, he has an outstanding track record in the biotech space, and for both these reasons, his hands-on involvement with the SciVac deal warrants attention.
SciVac’s flagship product is a Hepatitis B vaccine called Sci-B-Vac. Hepatitis B transfers from carrier to carrier through sexual contact, blood contamination and from mother to child. The virus gets into and replicates in hepatocytes, which are the primary functioning cell in the human liver. This invasion of hepatocyte cells leads to an immune response, during which the immune system attacks infected cells, causing damage not only to the infected cells, but also to wider liver function.
Currently, more than 170 countries have infant vaccination programs, and over 1 billion vaccines have been administered since 1982. The current standard of care necessitates three intravenous administrations over a period of six months. However, despite this vaccination effort, an astounding 2 billion people alive today have been infected with the virus at some point in their lives, an estimated 400 million people are currently carriers, 4 million individuals a year suffer from acute hepatitis B and 1.2 million people die every year from infection.
Infection rate is highest in central and southern Africa, Canada, the Middle East China and especially Southeast Asia where we see a higher than 8% prevalence. So if the current vaccination program is so widespread, why are there so many cases? First, there are a number of high-risk patients who don’t respond to the current vaccines, including sufferers of diabetes, cancer, HIV and renal disease. 50% of chronic liver disease sufferers of which there are 300 million globally, do not respond to current available vaccines. In addition, the current vaccinations are ineffective in the more than 2 million children born to infected mothers each year. Finally, with the current standard of care taking six months, frequent travelers are unprovided for.
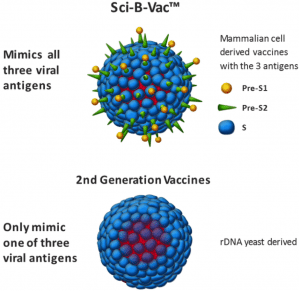
So what’s different about Sci-B-Vac? It is a third-generation vaccine, and it differs from the second-generation vaccine currently used in the vast majority of cases in that it contains three hepatitis B surface antigens. Surface antigens are the viral antigen mimics, which display to the human immune system and initiate the immune response. In the second-generation, the vaccine only displays one antigen called the “S” antigen. Sci-B-Vac mimics two further antigens – “preS1” and “preS2”. These extra displays improve vaccine response.
Image illustrating the difference between 2nd and 3rd generation HBV vaccines.
The third-generation vaccine has demonstrated efficacy in more than 20 company sponsored or investigator initiated trials across 5000+ individuals. The treatment is also already approved in 10 countries, and has been administered to more than 500,000 patients in these regions. Further, in two independent comparative studies, Sci-B-Vac demonstrated it produced seroprotection (i.e. protection from the virus the vaccination is designed to treat) more rapidly in both adults newborns than in healthy individuals treated with Engerix B – the current SOC vaccination.
This is all well and good, but why has Dr. Frost gotten involved? Perhaps because of the mitigated risks involved. The vaccine has already demonstrated safety and efficacy in 500,000 patients and is only approved in 10 countries. This gives us a clear path to phase 3 trials and US approval. As far as timeframes are concerned, SciVac expects to gain marketing approval for the vaccine in Latin America this year, while also meeting with the FDA and the BMA before the middle of 2016. The first quarter 2017, the company expects to have phase 3 enrolment complete in the US, and efficacy data for HIV and renal disease sufferers during first quarter 2018.
To put the market potential for these two indications into perspective, anywhere between 34 and 81% of 10 million chronic renal failure sufferers do not respond to Engerix B, and approximately 30% of 3 million HIV sufferers globally also do not respond. SciVac expects to gain approval for both of these indications in the US and Europe before the middle of 2018.
Hepatitis B is not the only liver disease Frost is involved in either. He is also involved in a hepatitis C vaccine company called Cocrystal Pharma, Inc. (COCP). Cocrystal is up 256% since August last year, down from gains of around 300% logged in April. Frost currently owns about 20% of Cocrystal individually through his investment trust, and his healthcare company Opko also has an 8% position – structuring its ownership in a very similar way to how Frost has set up in SciVac. Cocrystal itself went public through an acquisition structured in an almost identical fashion to SciVac, through an acquisition by BioZone Pharmaceuticals, Inc, then trading under the ticker “BZNE” and since renamed Cocrystal.
What’s even more interesting, is that there is speculation that we could see a further deal between Cocrystal and SciVac somewhere down the line. From a pipeline perspective, it seems to make sense, with both companies being involved in the hepatitis vaccination space. Additionally, from an ownership perspective, it looks to make even more sense – with Frost holding a high percentage stake in both.
Conclusion
Over the last couple of years Dr. Phillip Frost has racked up his holdings in the hepatitis vaccine space through two reverse merger public offerings, and one of them is up in the triple digit percentage range since his involvement. He has now cemented his involvement in the second, and with a risk-mitigated path to what looks like a high percentage chance of regulatory approval, we could see similar growth in SciVac. Of course, in development stage biotech, nothing is certain. However, if there is a man to keep an eye on in the space, it’s Frost.
**
Disclaimer:
Market Exclusive Is a financial portal geared to engaging discussion on current financial topics. Market Exclusive is not an investment advisor. Please read our full disclaimer at http://marketexclusive.com/about-us/disclaimer/