sponsored Content
Catalyst Biosciences Inc (NASDAQ:CBIO) shares were up over 15% at 11:00 AM ET on May 31, 2017, following the company’s announcement that the European Medicines Agency (EMA) Committee for Orphan Medicinal Products issued a positive opinion and recommended orphan medicinal product, or orphan drug, designation for CB 2679d/ISU304, for the treatment of hemophilia B.
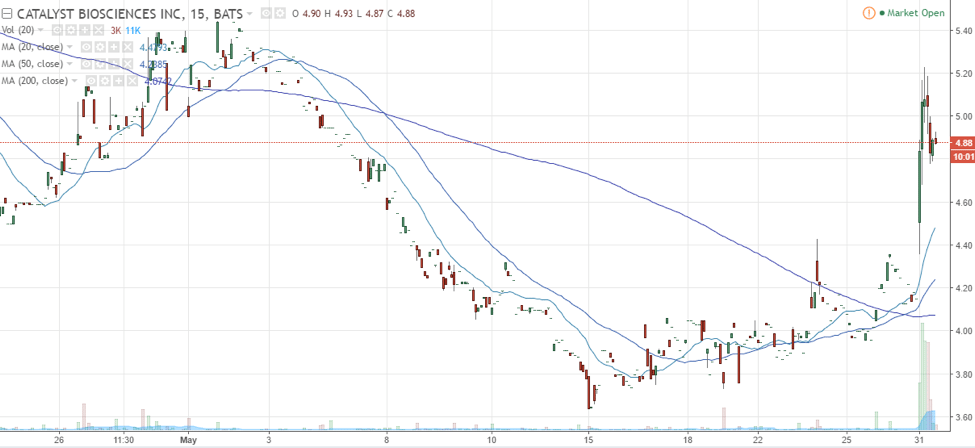
Here’s a look at Catalyst Biosciences following the announcement.
Source: TradingView
According to trader Jason Bond, “Generally, when we see micro-cap stocks report a positive catalyst, the stock price rises significantly. This was the case with Catalyst Biosciences. The company only has 4.26M shares outstanding and 4.10M shares floating. That in mind, since the stock has a low float, it could continue to see a significant rise, since some traders may demand more shares, on the long side, which should bid up the prices.”
Here’s a look at Catalyst Biosciences’ short interest metrics.
Source: Morningstar
Let’s take an in depth look into the catalyst that sent Catalyst Biosciences running higher recently. Typically, when we see a company receive orphan drug designation, shares rise significantly. Now, the currently approved therapies for the treatment of hemophilia B rely on intravenous infusions, and often, adherence and convenient access to peripheral veins is difficult to find. Therefore, these treatments often require the use of a central venous access device, which may increase the risks of infection, as well as thrombosis.
However, CB 2679d is a highly potent next-generation coagulation Factor IX variant, which has exhibited, in preclinical studies, the potential to normalize human Factor IX levels with a daily subcutaneous injection.
According President and Chief Executive Officer of Catalyst Biosciences Dr. Nassim Usman, “Obtaining orphan drug designation is an important part of our regulatory approval strategy for CB 2679d, and the receipt of the positive COMP opinion puts us one step closer to this goal.”
That in mind, traders viewed this orphan drug designation as a positive catalyst, which caused shares of CBIO to rise, and the stock could continue to build momentum off of this news.
Prior to this news, Catalyst Biosciences was down over 50% year to date, but this news could change the course for CBIO. Moreover, its first quarter 2017 quarterly earnings beat the consensus estimate recently. Let’s take a look at some highlights. According to Catalyst Biosciences, the company
- Raised $20.7 million through an underwritten public equity offering that included the full exercise of the underwriters’ over-allotment option to purchase additional shares and warrants on April 12, 2017
- Achieved key milestones with CB 2679d/ISU304, the Company’s next-generation coagulation Factor IX, as follows:
- Investigational New Drug (IND) application approved by the Korean Ministry of Food and Drug Safety (MFDS)
- Completion of IND-enabling toxicology studies triggered a milestone payment from Catalyst’s collaboration partner, ISU Abxis
- Advanced the development of marzeptacog alfa (activated), the Company’s next-generation Factor VIIa, including the following accomplishments:
- Received notice from the European Patent Office that the opposition period, for a patent granted to Catalyst, has expired and no opposition has been filed
- Selected a global contract research organization, INC Research, to conduct the Phase 2/3 efficacy clinical trial of marzeptacog alfa (activated) in individuals with hemophilia A or B with an inhibitor
That in mind, we’re going to want to watch data from Catalyst Biosciences data releases from its clinical trials, as well as its development pipeline.
This article is Sponsored Content