
Q: Can you please tell us about Todos and the specific problem or challenge that you are addressing?
Cancer is a disease that has touched all of our lives in some manner. What is known for certain is that early detection of the disease is critical. To this end, TODOS Medical is an innovative biotech company that is focused on creating a new methodology for cancer screening tests that make cancer detection more accurate, accessible, and affordable to the general public.
Early detection is key in saving lives. Todos will focus on the utilization of its tests for early detection and screening as its key market message. The test is low cost with no upfront equipment investment requirements, creating an economically sound method for early detection.
Corporate Profile:
Website: www.Todosmedical.com
Q: Can you please tell us a little about the market and the market opportunity?
While early detection of cancer is necessary if not vital, there are many obstacles to early detection. Some of these obstacles include high cost per screen, patient discomfort (mammogram, colonoscopy, MRI), accessibility, risk (radiation and invasive tests) and the need for medical specialists to interpret the test results. So from our perspective, there is a real need to mitigate and overcome the barriers that exist for the early detection of cancer. We at TODOS Medical, believe that we can offer screenings that provide faster results and at lower costs compared to the current competing technologies in today’s market.
Q: Please tell us a little about your technology?
Our Technology (TBIA) harnesses the immune system that detects cancer.
Our immune system detects cancer existence at early stages and attempts to kill it. Our TBIA technology recognizes and captures this biochemical event which acts as a “spectral fingerprint”.
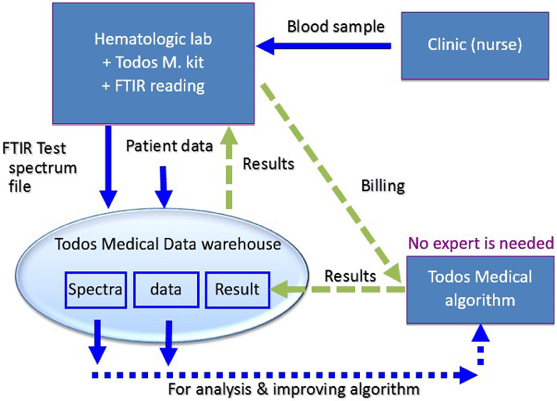
There is a scientific consensus that many different immune markers and biomarkers have to be taken into consideration to detect cancer existence. Our approach addresses this need since we check the total biochemical construction on the PBMC and plasma.
Q: What types of cancer is TODOS Medical concentrating on:
We are currently focusing our attention on cancer screenings for colon and breast cancer. However, we estimate that our methodology can be adapted to identify other types of cancer, and new products can be readily developed using our existing technology.
Q: What geographic markets are you focusing on currently?
TODOS Medical is based out of Rehovot, Israel. To date we have completed four clinical studies in Israel. We have also obtained the CE mark of approval from the European Union. In the near future we intend on continuing clinical studies in Israel and beginning studies in both Singapore and the United States.
Todos has growing clinical evidence supporting its colorectal and breast cancers screening tests with other tests in the pipeline. According to the American Cancer Society and Eurostat, the market for blood based colorectal cancer tests is 150 million tests in Europe and 87 million tests in the US, or $1.6 billion worldwide by 2019. While the market for breast cancer blood tests is 20 million tests in the US and 25 million tests in Europe, or $2.4 billion.
Q: What markets will you focus on in the near future and what is your plan to penetrate these markets?
TODOS Medical intends for its cancer screenings to be universally available. Todos will sell its test kits to private labs and lab chains, which provide testing services to patients, following medical referral. The test is designed as an early screening tool for cancer, enabling adoption for the general population. Todos opened an office in Singapore, we signed with a distributor covering parts of Eastern Europe and is putting efforts to establish relations in the US and Europe.
Q: What are the key benefits and features of your pipeline/drug for the target customer?
We propose a simple blood test for cancer screening, a test that is low cost and available for the mass population.
Q: Who are your competitors?
TODOS Medical has a number of competitors. These competitors include:
Current prevailing cancer screening tests utilize the standard procedures which, we believe, are typically uncomfortable, such as colonoscopy for colorectal cancer and mammography for breast cancer. In addition some of these tests have medium to low sensitivities/specificity and some have with adverse risks and are expensive. Furthermore, many of the existing screening methods depend on the technician’s or the physician’s capabilities, knowledge and interpretation. The existing screening methods usually detect cancer at later stages which is a key factor in saving lives, the earliest we detect the cancer the easier it is to cure it with lower costs.
In light of these drawbacks, we believe our competitive advantage is three-fold:
(i) the low cost of TBIA cancer screening tests
(ii) availability and accessibility to the mass population
(iii) good performances.
Q: What makes your pipeline stand out?
Blood based tests are the future of cancer screening. Todos has analyzed the existing limitations in solid tumor diagnosis and has developed its technology accordingly. TBIA is suitable for general market screening, projected to have high compliancy rates, due to its simplicity. The test is designed for general hematological labs, with no analysis requirement by cancer specialists, and does not require high capital equipment expense, enabling global adoption.
Q: Anything that you would like to add?
The company is working diligently to achieve its goal to provide cancer screening tests that makes early detection of cancer more accessible, and affordable to the general public.





